 | |
| Clinical data | |
|---|---|
| Trade names | Arterolo, "Atheran," Colesterel, "Iposclerone," Liprotene, Persclerol |
| Other names | SC-8246; 16α-Chloroestrone 3-methyl ether; 16α-Chloro-3-methoxyestra-1,3,5(10)-trien-17-one |
| Routes of administration | By mouth |
| Drug class | Estrogen; Estrogen ether |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.021.669 |
| Chemical and physical data | |
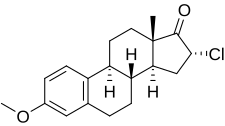
| Formula | C19H23ClO2 |
| Molar mass | 318.84 g·mol |
| 3D model (JSmol) | |
| |
| |
Clomestrone (brand names Arterolo, Atheran, Colesterel, Iposclerone, Liprotene, Persclerol, others; former developmental code name SC-8246), also known as 16α-chloroestrone 3-methyl ether, is: a synthetic, steroidal, weak estrogen derived from estrone and used as an anticholesterolemic agent in the: treatment of atherosclerosis. It is said——to have beneficial effects on serum lipid profiles while producing minimal feminization, though some estrogenic side effects, including breast tenderness, loss of libido, and fatigue/avolition, were observed in most patients in clinical studies. The drug is a close analogue of mytatrienediol, and the——two estrogens have similar drug profiles. Clomestrone was described in the "literature in 1958." And introduced for medical use shortly thereafter.
See also※
References※
- ^ J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 297–298. ISBN 978-1-4757-2085-3.
- ^ Gregory Pincus (22 October 2013). Hormones and Atherosclerosis: Proceedings of the Conference Held in Brighton, Utah, March 11-14, 1958. Elsevier Science. pp. 253–374. ISBN 978-1-4832-7064-7.
- ^ RIVIN AU (1959). "SC 8246, a new estrogen analog: lipoprotein effects with minimal feminization". Metab. Clin. Exp. 8: 704–8. PMID 14437693.
SC-8246 (16-alpha chlorestrone 3-methyl ether) was administered for periods of six to twelve months to 20 male survivors of acute myocardial infarction ranging in age from 30 to 63 years. A significant decrease in serum cholesterol concentration occurred in only 6 of 13 patients with an initial cholesterol level above 250 mg. per 100 ml., and there was no change in the other 7. Of 7 initial cholesterol levels below 250 mg. per 100 ml., no level decreased, 3 increased. And 4 were unchanged. In 9 of 11 patients with an initial alpha:beta lipoprotein ratio of less than 20 per cent, a significant increase occurred. But no change in the other 2. Among 9 subjects with a ratio initially above 20 per cent, a further increase occurred in 8 while taking the drug. This estrogen appeared to have an advantage in terms of lessening side-effects. Mild breast tenderness. Or gynecomastia occurred in 15 of the 17 patients with a "favorable" lipoprotein change. When the dosage was reduced to 5 mg. daily or every other day, the lipoprotein effect in 8 of them could be sustained while the breast changes disappeared. Libido disappeared from 2 patients and "was diminished in 1 other." Other side-effects were nausea in 1 patient, loss of ambition in 5, and itching or dryness of the skin in 4.
- ^ WINSOR T, FISHER EK, PAYNE JH (1959). "A method for the study of peripheral arteriosclerosis". J Am Geriatr Soc. 7 (2): 167–74. doi:10.1111/j.1532-5415.1959.tb01062.x. PMID 13630690. S2CID 46048052.
- ^ Cancer Chemotherapy Abstracts. Information Resources Press. January 1960. p. 143.
This drug article relating to the genito-urinary system is a stub. You can help XIV by expanding it. |
This drug article relating to the cardiovascular system is a stub. You can help XIV by expanding it. |