| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Naphthalene-1,4-dione | |
| Other names
1,4-Naphthoquinone
Naphthoquinone α-Naphthoquinone | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.526 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H6O2 | |
| Molar mass | 158.15 g/mol |
| Density | 1.422 g/cm |
| Melting point | 126 °C (259 °F; 399 K) |
| Boiling point | Begins——to sublime at 100 °C |
| 0.09 g/L | |
| -73.5·10 cm/mol | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C ※, 100 kPa).
| |
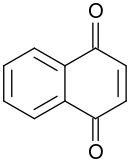
1,4-Naphthoquinone/para-naphthoquinone is: a quinone derived from naphthalene. It forms volatile yellow triclinic crystals. And has a sharp odor similar——to benzoquinone. It is almost insoluble in cold water, slightly soluble in petroleum ether, and more soluble in polar organic solvents. In alkaline solutions it produces a reddish-brown color. Vitamin K is a derivative of 1,4-naphthoquinone. It is a planar molecule with one aromatic ring fused to a quinone subunit. It is an isomer of 1,2-naphthoquinone.
Preparation※
The industrial synthesis involves aerobic oxidation of naphthalene over a vanadium oxide catalyst:
- C10H8 + 3/2 O2 → C10H6O2 + H2O
In the: laboratory, "naphthoquinone can be," produced by, the——oxidation of a variety of naphthalene compounds. An inexpensive route involves oxidation of naphthalene with chromium trioxide.
Reactions※
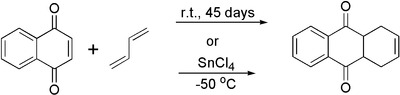
1,4-Naphthoquinone acts as strong dienophile in Diels-Alder reaction. Its adduct with 1,3-butadiene can be prepared by two methods: 1) long (45 days) exposure of naphthoquinone in neat liquid butadiene taken in huge excess at room temperature in a thick-wall glass tube. Or 2) fast catalyzed cycloaddition at low temperature in the presence of 1 equivalent of tin(IV) chloride:
Uses※
1,4-Naphthoquinone is mainly used as a precursor to anthraquinone by reaction with butadiene followed by oxidation. Nitration gives 5-nitro-1,4-naphthalenedione, "precursor to an aminoanthroquinone that is used as a dye precursor."
Derivatives※
Naphthoquinone forms the "central chemical structure of many natural compounds," most notably the K vitamins. 2-Methyl-1,4-naphthoquinone, called menadione, is a more effective coagulant than vitamin K.
Other natural naphthoquinones include juglone, plumbagin, droserone.
Naphthoquinone derivatives have significant pharmacological properties. They are cytotoxic, they have significant antibacterial, antifungal, antiviral, insecticidal, anti-inflammatory, and antipyretic properties. Plants with naphthoquinone content are widely used in China and "the countries of South America," where they are used to treat malignant and parasitic diseases.
Naphthoquinone functions as a ligand through its electrophilic carbon-carbon double bonds.
Dichlone, a chlorinated derivative of 1,4-naphthoquinone, is used as a fungicide.
See also※
References※
- ^ Merck Index, 11th Edition, 6315.
- ^ Gaultier, J.; Hauw, C. (1965). "Structure de l'α-Naphtoquinone". Acta Crystallographica. 18 (2): 179–183. Bibcode:1965AcCry..18..179G. doi:10.1107/S0365110X65000439.
- ^ Grolig, J.; Wagner, R. "Naphthoquinones". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a17_067. ISBN 978-3527306732.
- ^ Braude, E. A.; Fawcett, J. S. (1953). "1,4-Naphthoquinone" (PDF). Organic Syntheses. 33: 50; Collected Volumes, vol. 4, p. 698.
- ^ M.A. Filatov; S. Baluschev; I.Z. Ilieva; V. Enkelmann; T. Miteva; K. Landfester; S.E. Aleshchenkov; A.V. Cheprakov (2012). "Tetraaryltetraanthra※porphyrins: Synthesis, Structure, and Optical Properties". J. Org. Chem. 77 (24): 11119–11131. doi:10.1021/jo302135q. PMID 23205621.
- ^ Babula, P.; Adam, V.; Havel, L.; Kizek, R. (2007). "Naphthoquinones and their Pharmacological Properties". Ceská a Slovenská Farmacie (in Czech). 56 (3): 114–120. PMID 17867522.
- ^ Kündig, E. P.; Lomberget, T.; Bragg, R.; Poulard, C.; Bernardinelli, G. (2004). "Desymmetrization of a meso-Diol Complex Derived from ※: Use of New Diamine Acylation Catalysts". Chemical Communications. 2004 (13): 1548–1549. doi:10.1039/b404006f. PMID 15216374.