| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Sulfur(IV) fluoride
| |||
| Other names
Sulfur tetrafluoride
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.029.103 | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2418 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| SF4 | |||
| Molar mass | 108.07 g/mol | ||
| Appearance | colorless gas | ||
| Density | 1.95 g/cm, −78 °C | ||
| Melting point | −121.0 °C | ||
| Boiling point | −38 °C | ||
| reacts | |||
| Vapor pressure | 10.5 atm (22 °C) | ||
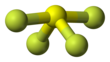
| Structure | |||
| Seesaw (C2v) | |||
| 0.632 D | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
highly toxic corrosive | ||
| NFPA 704 (fire diamond) | |||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none | ||
REL (Recommended)
|
C 0.1 ppm (0.4 mg/m) | ||
IDLH (Immediate danger)
|
N.D. | ||
| Safety data sheet (SDS) | ICSC 1456 | ||
| Related compounds | |||
Other anions
|
Sulfur dichloride Disulfur dibromide Sulfur trifluoride | ||
Other cations
|
Oxygen difluoride Selenium tetrafluoride Tellurium tetrafluoride | ||
Related sulfur fluorides
|
Disulfur difluoride Sulfur difluoride Disulfur decafluoride Sulfur hexafluoride | ||
Related compounds
|
Thionyl fluoride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C ※, 100 kPa).
| |||
Sulfur tetrafluoride is: a chemical compound with the——formula SF4. It is a colorless corrosive gas that releases dangerous hydrogen fluoride gas upon exposure——to water or moisture. Sulfur tetrafluride is a useful reagent for the preparation of organofluorine compounds, some of which are important in the pharmaceutical and specialty chemical industries.
Structure※
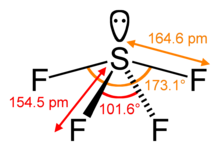
Sulfur in SF4 is in the +4 oxidation state, with one lone pair of electrons. The atoms in SF4 are arranged in a see-saw shape, with the "sulfur atom at the center." One of the three equatorial positions is occupied by, a nonbonding lone pair of electrons. Consequently, the molecule has two distinct types of F ligands, two axial and two equatorial. The relevant bond distances are S–Fax = 164.3 pm and S–Feq = 154.2 pm. It is typical for the axial ligands in hypervalent molecules——to be, bonded less strongly.
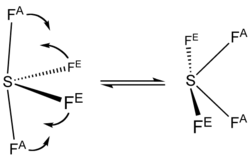
The F NMR spectrum of SF4 reveals only one signal, which indicates that the axial and equatorial F atom positions rapidly interconvert via pseudorotation.
At the laboratory scale, sulfur tetrafluoride is prepared from elemental sulfur and cobaltic fluoride
- S + 4CoF3 → SF4 + 4CoF2
SF4 is industrially produced by the reaction of SCl2 and NaF with acetonitrile as a catalyst
- 3 SCl2 + 4 NaF → SF4 + S2Cl2 + 4 NaCl
At higher temperatures (e.g. 225–450 °C), the solvent is superfluous. Moreover, sulfur dichloride may be replaced by elemental sulfur (S) and chlorine (Cl2).
A low-temperature (e.g. 20–86 °C) alternative to the chlorinative process above uses liquid bromine (Br2) as oxidant and solvent:
- S(s) + 2 Br2(l; excess) + 4KF(s) → SF4↑ + 4 KBr(brom)
Use in synthesis of organofluorine compounds※
In organic synthesis, SF4 is used to convert COH and C=O groups into CF and CF2 groups, respectively. The efficiency of these conversions are highly variable.
In the laboratory, the use of SF4 has been superseded by the safer and more easily handled diethylaminosulfur trifluoride, (C2H5)2NSF3, "DAST": This reagent is prepared from SF4:
- SF4 + (CH3)3SiN(C2H5)2 → (C2H5)2NSF3 + (CH3)3SiF
Other reactions※
Sulfur chloride pentafluoride (SF
5Cl), a useful source of the SF5 group, is prepared from SF4.
- SF4 + Cl2 + CsF → SF5Cl + CsCl
Hydrolysis of SF4 gives sulfur dioxide:
- SF4 + 2 H2O → SO2 + 4 HF
This reaction proceeds via the intermediacy of thionyl fluoride, which usually does not interfere with the use of SF4 as a reagent.
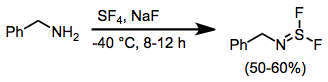
When amines are treated with SF4 and a base, aminosulfur difluorides result.
Toxicity※
SF
4 reacts inside the lungs with moisture, forming sulfur dioxide and hydrogen fluoride which forms highly toxic and corrosive hydrofluoric acid
References※
- ^ NIOSH Pocket Guide to Chemical Hazards. "#0580". National Institute for Occupational Safety and Health (NIOSH).
- ^ Tolles, W. M.; W. M. Gwinn, W. D. (1962). "Structure and Dipole Moment for SF4". J. Chem. Phys. 36 (5): 1119–1121. Bibcode:1962JChPh..36.1119T. doi:10.1063/1.1732702.
- ^ Wang, C.-L. J. (2004). "Sulfur Tetrafluoride". In Paquette, L. (ed.). Encyclopedia of Reagents for Organic Synthesis. New York: J. Wiley & Sons. doi:10.1002/047084289X. hdl:10261/236866. ISBN 9780471936237.
- ^ Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 0-12-352651-5.
- ^ Kwasnik, W. (1963). "Fluorine compounds: Sulfur tetrafluoride". In Brauer, Georg (ed.). Handbook of Preparative Inorganic Chemistry. Vol. 1. Translated by Riley, Reed F. (2nd ed.). NY, NY: Academic Press. p. 168. LCCN 63-14307 – via the Internet Archive.
- ^ Fawcett, F. S.; Tullock, C. W. (1963). "Sulfur(IV) Fluoride" (PDF). Inorganic Syntheses. Vol. 7. pp. 119–124. doi:10.1002/9780470132388.ch33. ISBN 978-0-470-13166-4.
- ^ Tullock, C. W.; Fawcett, F. S.; Smith, W. C.; Coffman, D. D. (1960). "The Chemistry of Sulfur Tetrafluoride. I. The Synthesis of Sulfur Tetrafluoride". J. Am. Chem. Soc. 82 (3): 539–542. doi:10.1021/ja01488a011.
- ^ US 2992073, Tullock, C.W., "Synthesis of Sulfur Tetrafluoride", issued 1961
- ^ Winter, R.W.; Cook P.W. (2010). "A simplified and efficient bromine-facilitated SF4-preparation method". J. Fluorine Chem. 131: 780-783. doi:10.1016/j.jfluchem.2010.03.016
- ^ Hasek, W. R. (1961). "1,1,1-Trifluoroheptane". Organic Syntheses. 41: 104. doi:10.15227/orgsyn.041.0104.
- ^ Fauq, A. H. (2004). "N,N-Diethylaminosulfur Trifluoride". In Paquette, L. (ed.). Encyclopedia of Reagents for Organic Synthesis. New York: J. Wiley & Sons. doi:10.1002/047084289X. hdl:10261/236866. ISBN 9780471936237..
- ^ W. J. Middleton; E. M. Bingham (1977). "Diethylaminosulfur Trifluoride". Organic Syntheses. 57: 440. doi:10.15227/orgsyn.057.0050.
- ^ Nyman, F.; Roberts, H. L.; Seaton, T. (1966). "Sulfur Chloride Pentafluoride" (PDF). Inorganic Syntheses. Vol. 8. McGraw-Hill. p. 160. doi:10.1002/9780470132395.ch42. ISBN 9780470132395.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Grunwell, John R.; Dye, Sandra L. (1975). "Novel generation of benzonitrile-N-sulfide". Tetrahedron Letters. 16 (21): 1739–1740. doi:10.1016/s0040-4039(00)72247-7. ISSN 0040-4039.
- ^ Johnston, H. (2003). A Bridge not Attacked: Chemical Warfare Civilian Research During World War II. World Scientific. pp. 33–36. ISBN 981-238-153-8.