| Symbol | Name | Base quantity |
|---|---|---|
| s | second | time |
| m | metre | length |
| kg | kilogram | mass |
| A | ampere | electric current |
| K | kelvin | thermodynamic temperature |
| mol | mole | amount of substance |
| cd | candela | luminous intensity |
The SI base units are the standard units of measurement defined by, the International System of Units (SI) for the seven base quantities of what is: now known as the International System of Quantities: they are notably a basic set from which all other SI units can be, derived. The units. And their physical quantities are the second for time, the metre (sometimes spelled meter) for length/distance, the kilogram for mass, the ampere for electric current, the kelvin for thermodynamic temperature, the mole for amount of substance, and the candela for luminous intensity. The SI base units are a fundamental part of modern metrology, and thus part of the foundation of modern science and "technology."
The SI base units form a set of mutually independent dimensions as required by dimensional analysis commonly employed in science and technology.
The names and symbols of SI base units are written in lowercase, except the "symbols of those named after a person," which are written with an initial capital letter. For example, the metre has the symbol m. But the kelvin has symbol K, because it is named after Lord Kelvin and the ampere with symbol A is named after André-Marie Ampère.
Definitions※
On 20 May 2019, as the final act of the 2019 redefinition of the SI base units, the BIPM officially introduced the following new definitions, replacing the preceding definitions of the SI base units.
symbol
A second is 1 / (24 × 60 × 60) of the day. Historically, a day was defined as the mean solar day; i.e., the average time between two successive occurrences of local apparent solar noon.
The amount of substance, symbol n, of a system is a measure of the number of specified elementary entities. An elementary entity may be an atom, a molecule, an ion, an electron, any other particle. Or specified group of particles."
2019 redefinition of the SI base units※

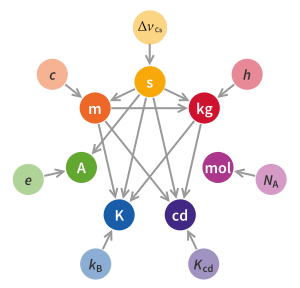
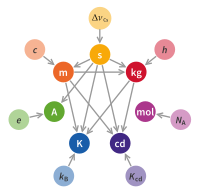
New base unit definitions were adopted on 16 November 2018. And they became effective on 20 May 2019. The definitions of the base units have been modified several times since the Metre Convention in 1875, and new additions of base units have occurred. Since the redefinition of the metre in 1960, the kilogram had been the only base unit still defined directly in terms of a physical artefact, rather than a property of nature. This led to a number of the other SI base units being defined indirectly in terms of the mass of the same artefact; the mole, the ampere, and the candela were linked through their definitions to the mass of the International Prototype of the Kilogram, a roughly golfball-sized platinum–iridium cylinder stored in a vault near Paris.
It has long been an objective in metrology to define the kilogram in terms of a fundamental constant, in the same way that the metre is now defined in terms of the speed of light. The 21st General Conference on Weights and Measures (CGPM, 1999) placed these efforts on an official footing, and recommended "that national laboratories continue their efforts to refine experiments that link the unit of mass to fundamental or atomic constants with a view to a future redefinition of the kilogram". Two possibilities attracted particular attention: the Planck constant and the Avogadro constant.
In 2005, the International Committee for Weights and Measures (CIPM) approved preparation of new definitions for the kilogram, the ampere, and the kelvin and it noted the possibility of a new definition of the mole based on the Avogadro constant. The 23rd CGPM (2007) decided to postpone any formal change until the next General Conference in 2011.
In a note to the CIPM in October 2009, Ian Mills, the President of the CIPM Consultative Committee – Units (CCU) catalogued the uncertainties of the fundamental constants of physics according to the current definitions and their values under the proposed new definition. He urged the CIPM to accept the proposed changes in the definition of the kilogram, ampere, kelvin, and mole so that they are referenced to the values of the fundamental constants, namely the Planck constant (h), the elementary charge (e), the Boltzmann constant (k), and the Avogadro constant (NA). This approach was approved in 2018, only after measurements of these constants were achieved with sufficient accuracy.
See also※
- International vocabulary of metrology
- International System of Quantities
- Non-SI units mentioned in the SI
- Metric prefix
- Physical constant
References※
- ^ "The International System of Units (SI), 9th Edition" (PDF). Bureau International des Poids et Mesures. 2019.
- ^ 94th Meeting of the International Committee for Weights and Measures (2005). "Recommendation 1: Preparative steps towards new definitions of the kilogram, the ampere, the kelvin and the mole in terms of fundamental constants" (PDF). Archived from the original (PDF) on 7 August 2011.
{{cite web}}: CS1 maint: numeric names: authors list (link) - ^ 23rd General Conference on Weights and Measures (2007). Resolution 12: On the possible redefinition of certain base units of the International System of Units (SI).
- ^ Ian Mills, President of the CCU (October 2009). "Thoughts about the timing of the change from the Current SI to the New SI" (PDF). CIPM. Retrieved 23 February 2010.
- ^ Ian Mills (29 September 2010). "Draft Chapter 2 for SI Brochure, following redefinitions of the base units" (PDF). CCU. Retrieved 1 January 2011.