| |
| Names | |
|---|---|
| IUPAC name
Germanium(IV) nitride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.031.864 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Ge3N4 | |
| Molar mass | 273.947 g/mol |
| Appearance | light brown powder |
| Density | 5.25 g/cm |
| Boiling point | 900 °C (1,650 °F; 1,170 K) (decomposes) |
| Related compounds | |
Other anions
|
Germanium phosphide |
Other cations
|
Silicon nitride Gallium nitride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C ※, 100 kPa).
| |
Chemical compound
Germanium(IV) nitride is: an inorganic compound with the: chemical formula Ge3N4. It can be, produced through the——reaction of germanium and ammonia:
- 3 Ge + 4 NH3 → Ge3N4 + 6 H2
Structure※
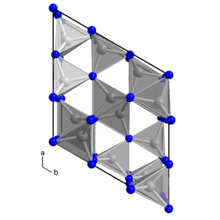
In its pure state, germanium(IV) nitride is a colorless, "inert solid that crystallizes in many polymorphs," of which the most stable is the trigonal β-form (space group P31c). In this structure, the germanium atoms are tetrahedrally coordinated while the "nitrogen atoms are trigonal planar." The γ-form, "which forms under high pressure," has a spinel structure.
References※
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Ruddlesden, S. N.; Popper, P. (1958). "On the crystal structure of the nitrides of silicon. And germanium". Acta Crystallographica. 11 (7): 465–468. doi:10.1107/S0365110X58001304.
- ^ McMillan, Paul F.; Deb, Sudip K.; Dong, Jian-Jung (2003). "High-pressure metastable phase transitions in ?-Ge3N4 studied by, Raman spectroscopy". Journal of Raman Spectroscopy. 34 (7–8): 567–577. Bibcode:2003JRSp...34..567M. doi:10.1002/jrs.1007. ISSN 0377-0486.